Multiple Covalent Triazine Frameworks with Strong Polysulfide Chemisorption for Enhanced Lithium‐Sulfur Batteries - Wang - 2019 - ChemElectroChem - Wiley Online Library
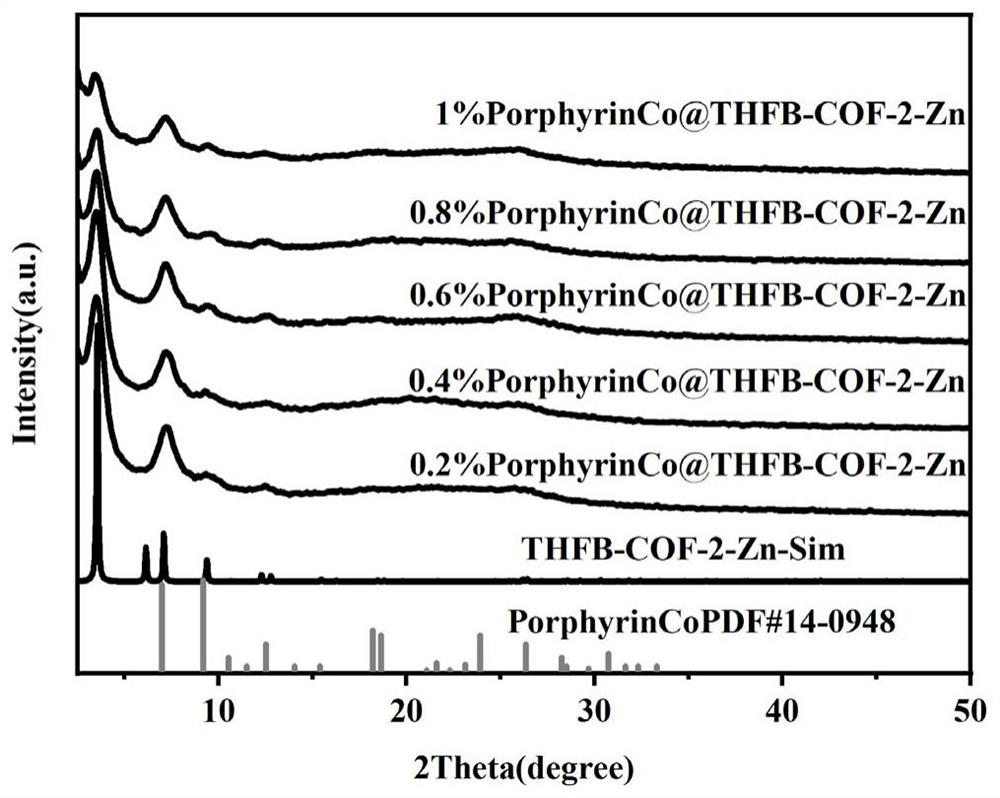
Preparation of triazine ring covalent organic framework material doped porphyrin cobalt photocatalyst - Eureka | Patsnap

From Triazines to Heptazines: Deciphering the Local Structure of Amorphous Nitrogen-Rich Carbon Nitride Materials | Journal of the American Chemical Society

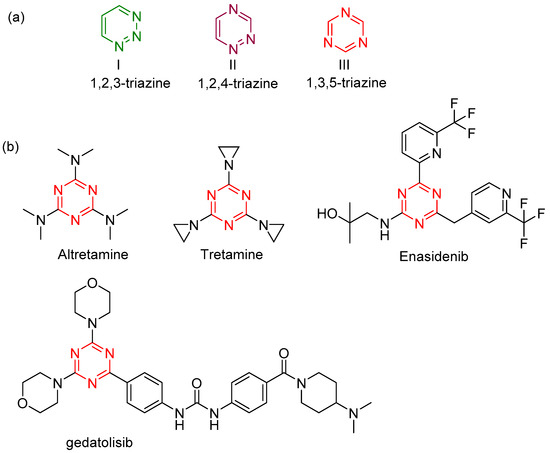
1,3,5‐Triazine: A versatile pharmacophore with diverse biological activities - Singh - 2021 - Archiv der Pharmazie - Wiley Online Library
![OPENING OF THE 1,3,5-TRIAZINE RING IN 3-METHYL-5-(TRINITROMETHYL)TETRAZOLO[1,5-<i>a</i>][1,3,5]TRIAZIN-7-ONE BY THE ACTION OF ALCOHOLS | Бахарев | Chemistry of Heterocyclic Compounds OPENING OF THE 1,3,5-TRIAZINE RING IN 3-METHYL-5-(TRINITROMETHYL)TETRAZOLO[1,5-<i>a</i>][1,3,5]TRIAZIN-7-ONE BY THE ACTION OF ALCOHOLS | Бахарев | Chemistry of Heterocyclic Compounds](https://hgs.osi.lv/public/journals/1/cover_article_6538_en_US.png)
OPENING OF THE 1,3,5-TRIAZINE RING IN 3-METHYL-5-(TRINITROMETHYL)TETRAZOLO[1,5-<i>a</i>][1,3,5]TRIAZIN-7-ONE BY THE ACTION OF ALCOHOLS | Бахарев | Chemistry of Heterocyclic Compounds

1, 3, 5-Triazine and 2, 4, 6-trichloro-1, 3, 5-triazine as Antimicrobial Drugs | Indian Health Journal
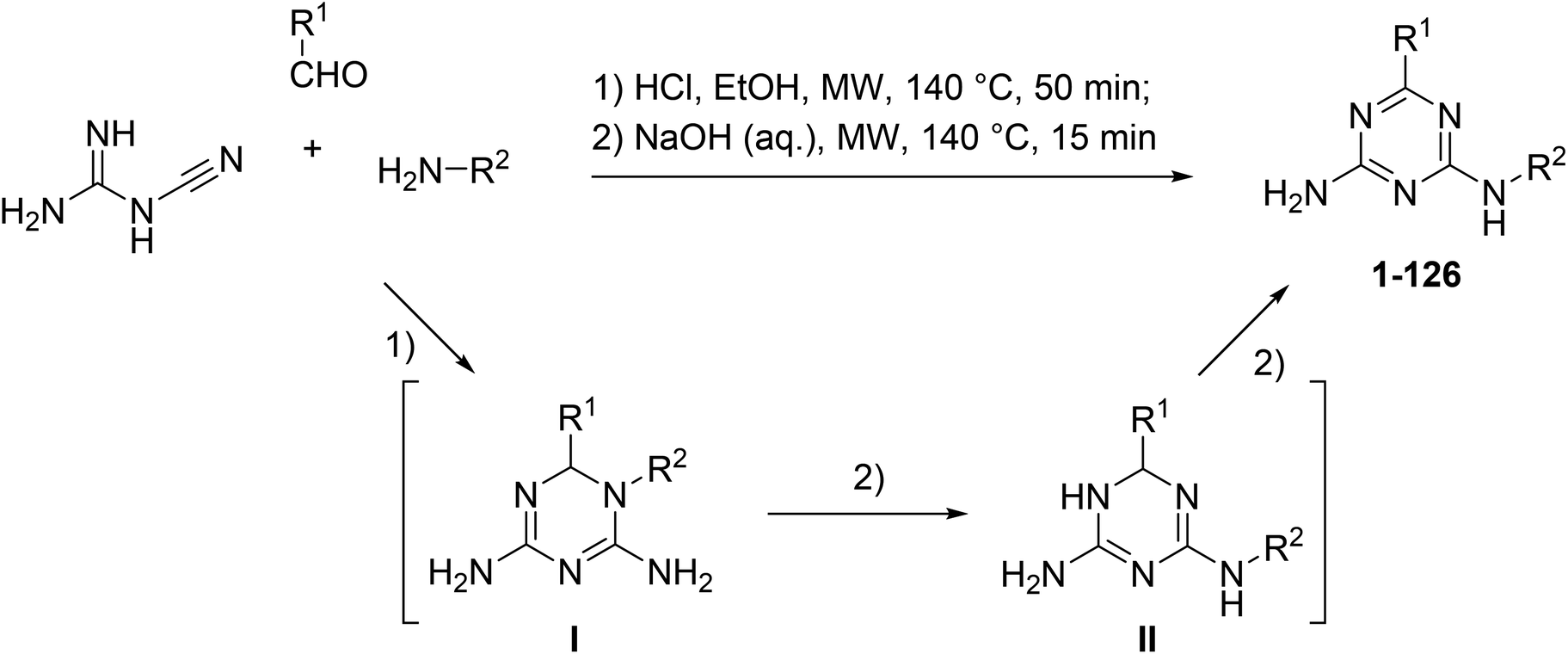
6, N 2 -Diaryl-1,3,5-triazine-2,4-diamines: synthesis, antiproliferative activity and 3D-QSAR modeling - RSC Advances (RSC Publishing) DOI:10.1039/D0RA00643B
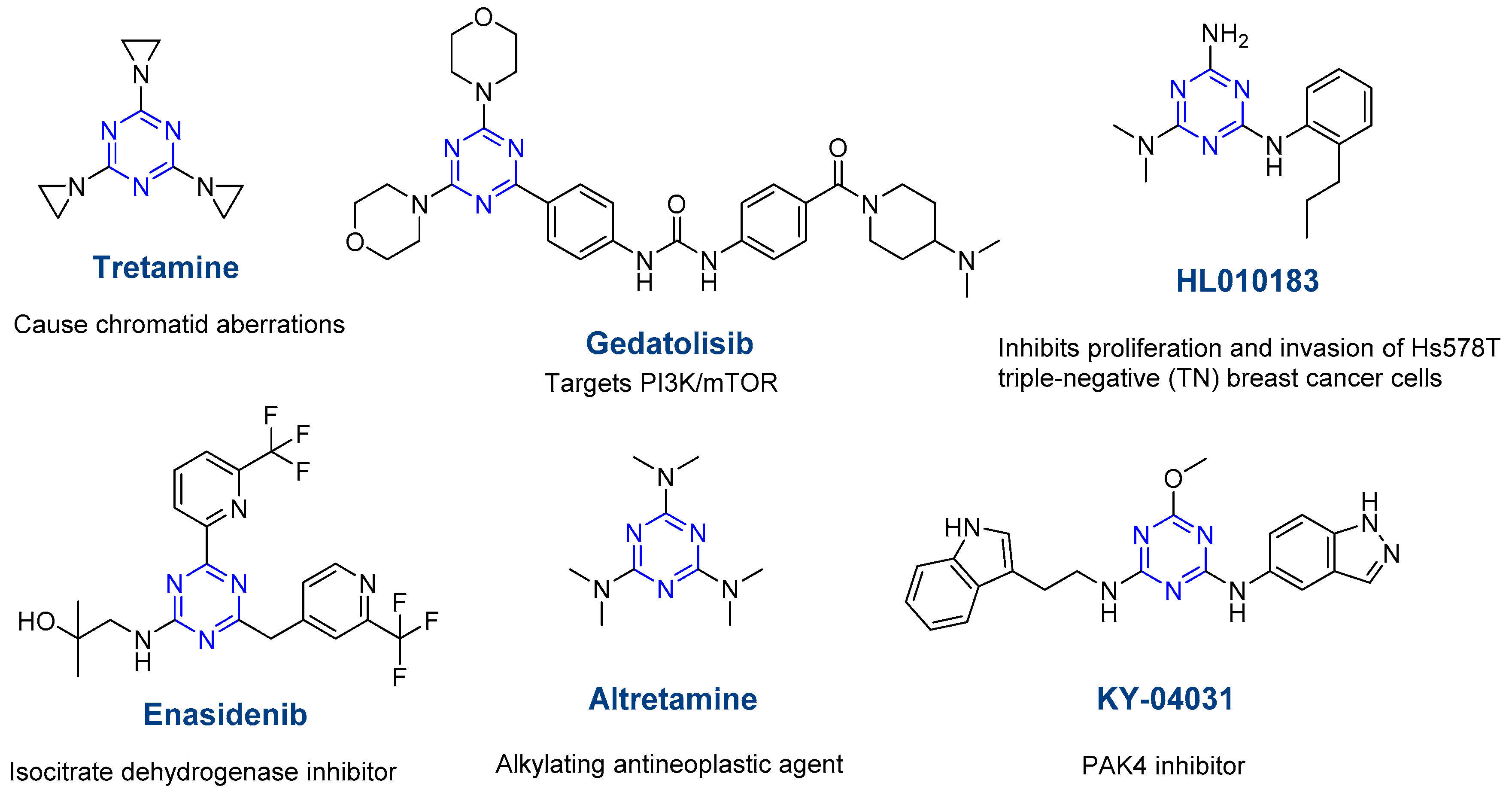
Molecules | Free Full-Text | s-Triazine: A Privileged Structure for Drug Discovery and Bioconjugation
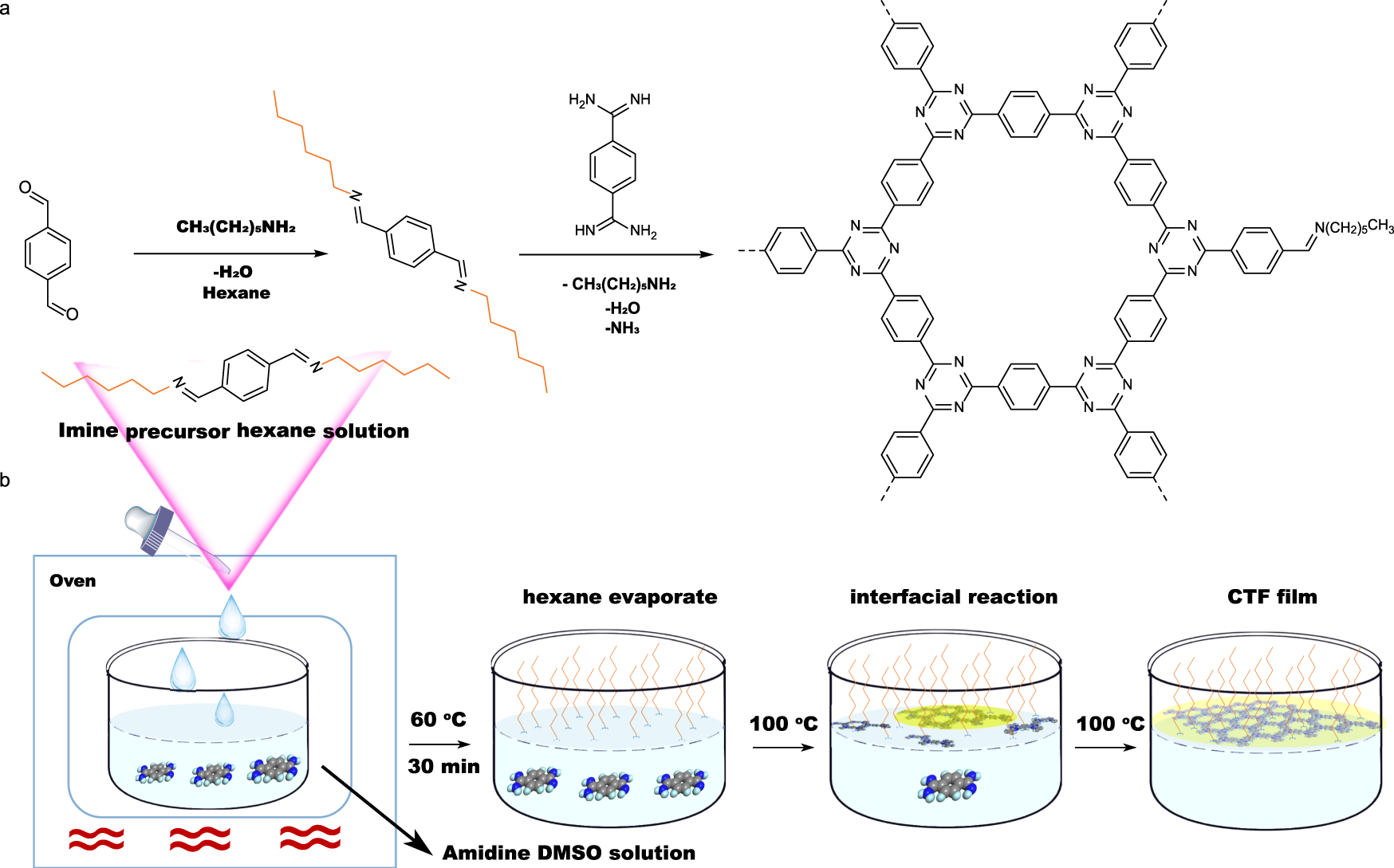
Immobilized covalent triazine frameworks films as effective photocatalysts for hydrogen evolution reaction | Nature Communications


![PDF] Synthesis of 2,4,6-tri-substituted-1,3,5-triazines. | Semantic Scholar PDF] Synthesis of 2,4,6-tri-substituted-1,3,5-triazines. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/72dd0b1e535d3ae5949e834703738b8b3d7ef130/2-Figure1-1.png)
![Triazine ring structure after self-polymerization [8] | Download Scientific Diagram Triazine ring structure after self-polymerization [8] | Download Scientific Diagram](https://www.researchgate.net/publication/371952688/figure/fig2/AS:11431281171244168@1688089831192/Triazine-ring-structure-after-self-polymerization-8.png)