A novel route to substituted poly(vinyl pyrrolidone)s via simple functionalization of 1-vinyl-2-pyrrolidone in the 3-position by ring-opening reactions - ScienceDirect

a) Energy diagram of mechanism for formation of pyrrolidine ring from... | Download Scientific Diagram
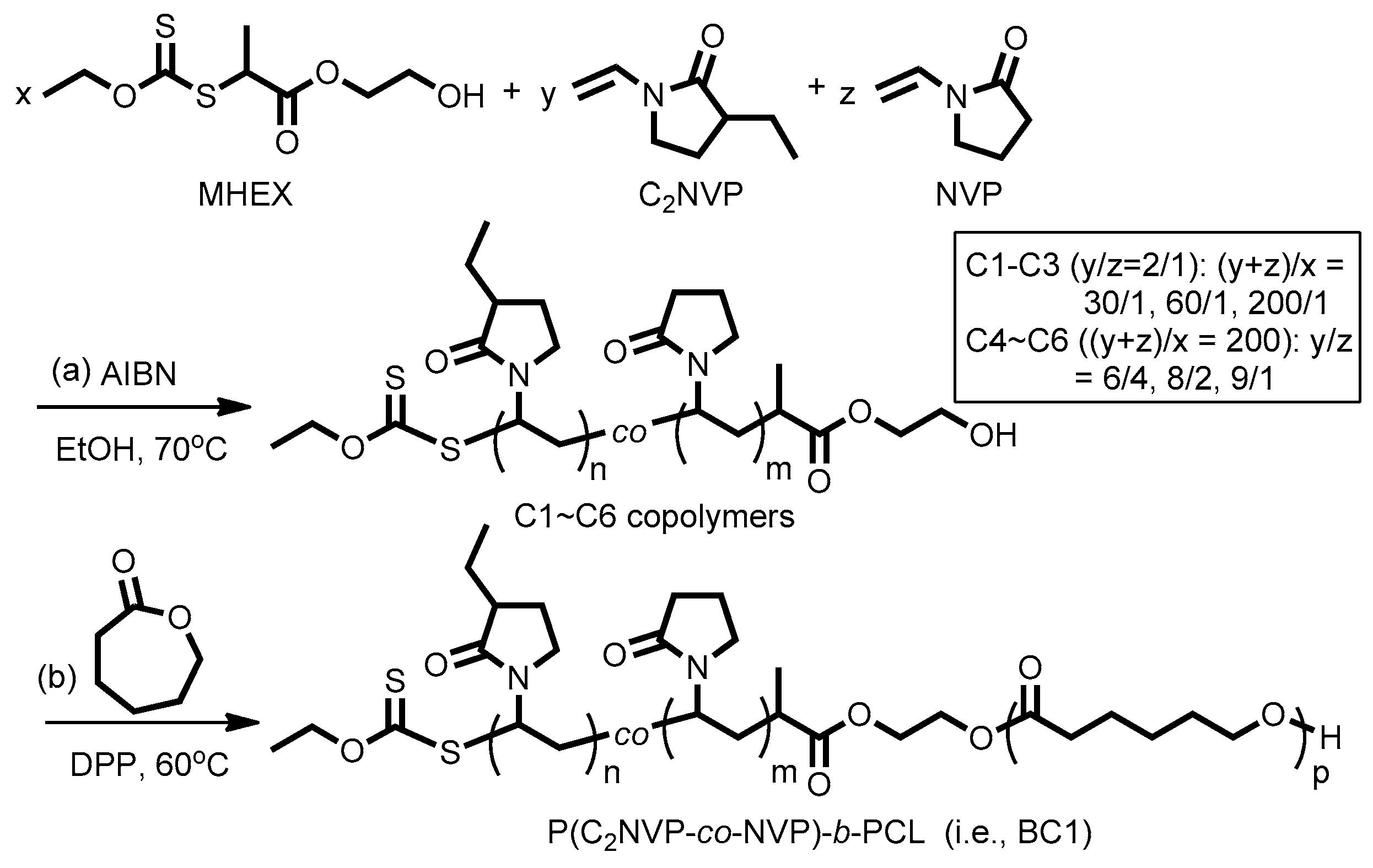
Polymers | Free Full-Text | Synthesis of PNVP-Based Copolymers with Tunable Thermosensitivity by Sequential Reversible Addition–Fragmentation Chain Transfer Copolymerization and Ring-Opening Polymerization

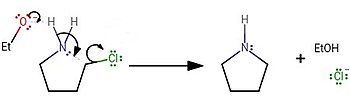
A Mild and Efficient Procedure for Ring-Opening Reactions of Piperidine and Pyrrolidine Derivatives by Single Electron Transfer Photooxidation - ScienceDirect

Suspension polymerization of 2‐pyrrolidone in the presence of CO2 and organic promoters - Chen - 2021 - Journal of Applied Polymer Science - Wiley Online Library

A Mild and Efficient Procedure for Ring-Opening Reactions of Piperidine and Pyrrolidine Derivatives by Single Electron Transfer Photooxidation - ScienceDirect
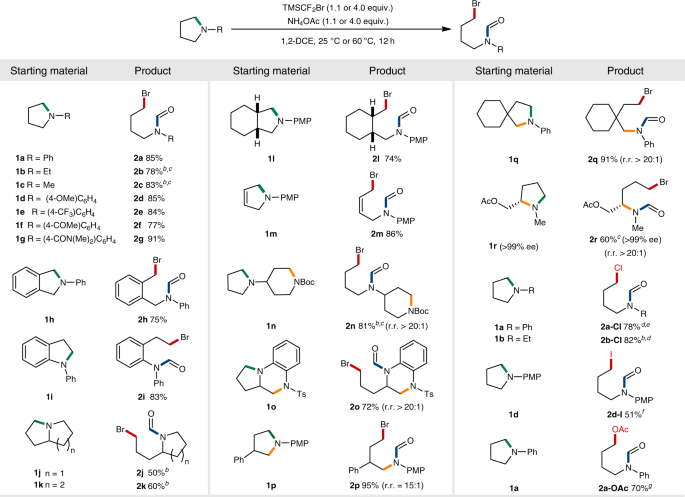
Ring-opening functionalizations of unstrained cyclic amines enabled by difluorocarbene transfer | Nature Communications

Iron-Catalyzed Transfer Hydrogenation in Aged N-Methyl-2-pyrrolidone: Reductive Ring-Opening of 3,5-Disubstituted Isoxazoles and Isoxazolines | The Journal of Organic Chemistry

Numbering of pyrrolidine's ring in N-acetylproline. The bond-cutting... | Download Scientific Diagram

Iron-Catalyzed Transfer Hydrogenation in Aged N-Methyl-2-pyrrolidone: Reductive Ring-Opening of 3,5-Disubstituted Isoxazoles and Isoxazolines | The Journal of Organic Chemistry

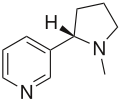
Molecules | Free Full-Text | Recent Advances in Asymmetric Synthesis of Pyrrolidine-Based Organocatalysts and Their Application: A 15-Year Update

Physicochemical Characterization of Polyvinyl Pyrrolidone: A Tale of Two Polyvinyl Pyrrolidones | ACS Omega

Synthesis of biobased N-substituted pyrrolidone ring-based polyamides... | Download Scientific Diagram
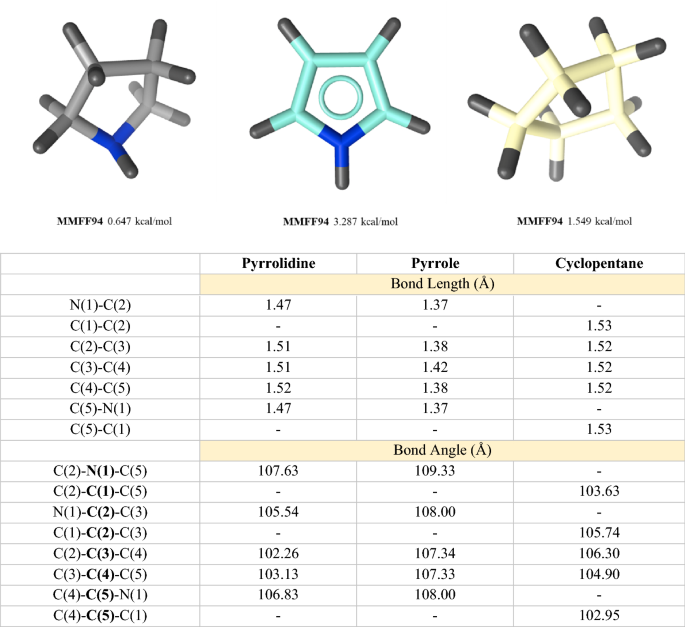
Pyrrolidine in Drug Discovery: A Versatile Scaffold for Novel Biologically Active Compounds | SpringerLink
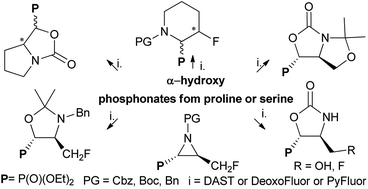
Pyrrolidine and oxazolidine ring transformations in proline and serine derivatives of α-hydroxyphosphonates induced by deoxyfluorinating reagents - RSC Advances (RSC Publishing)