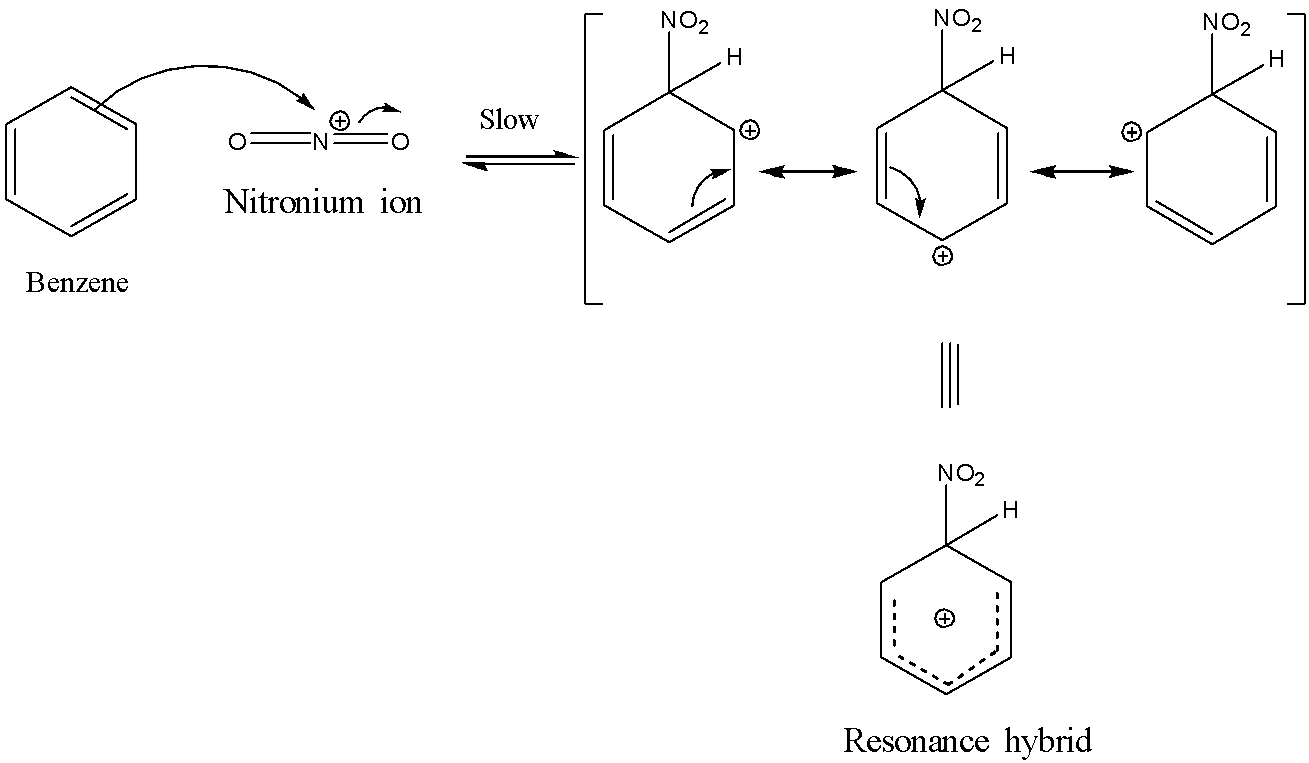
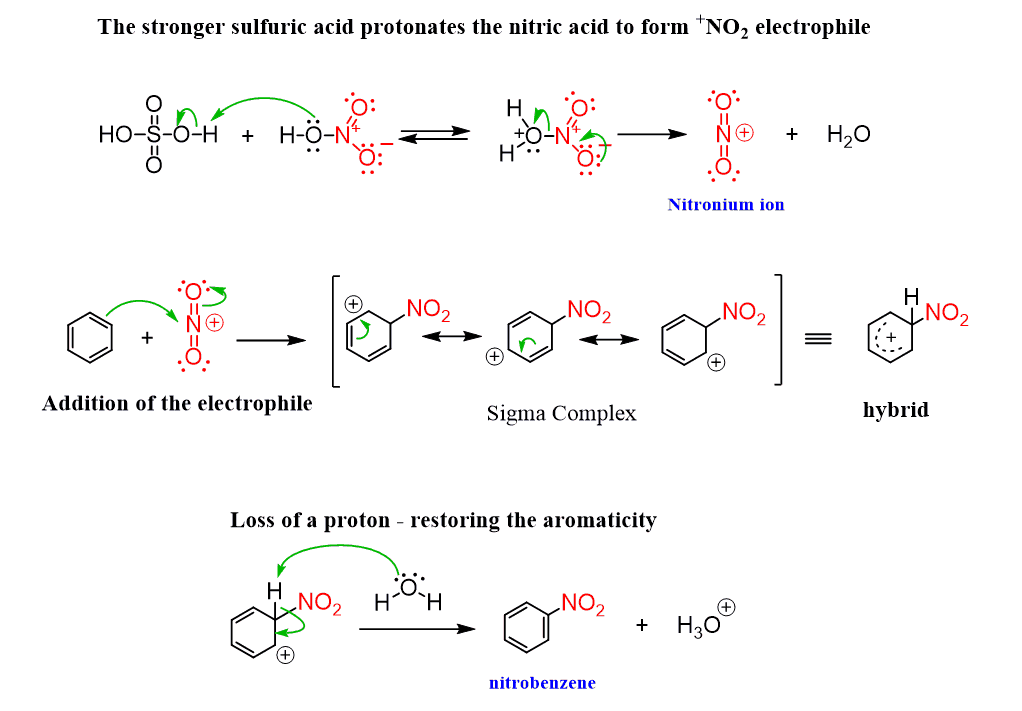
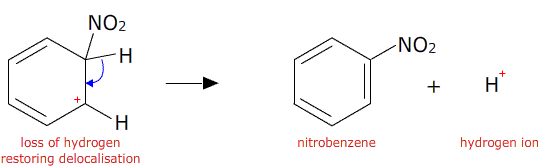
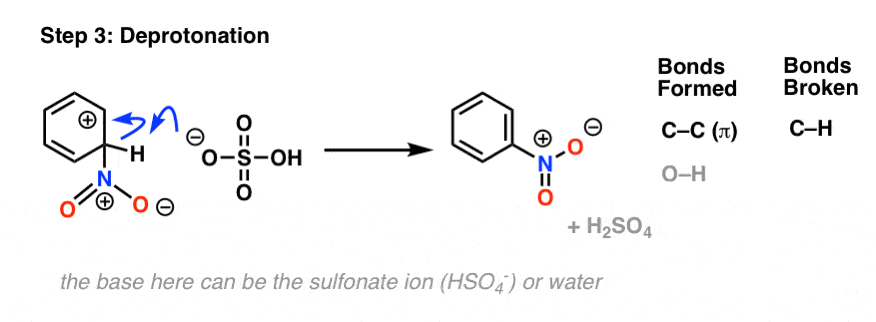
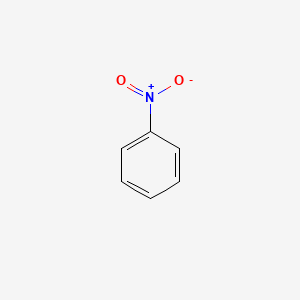
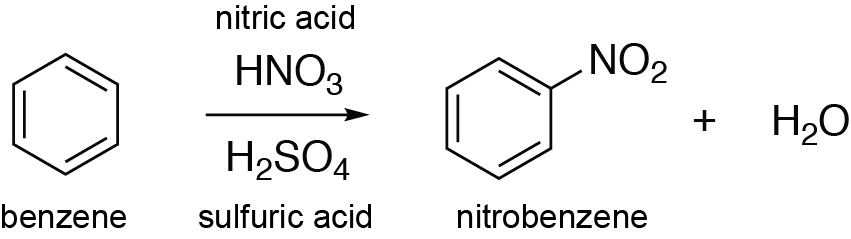
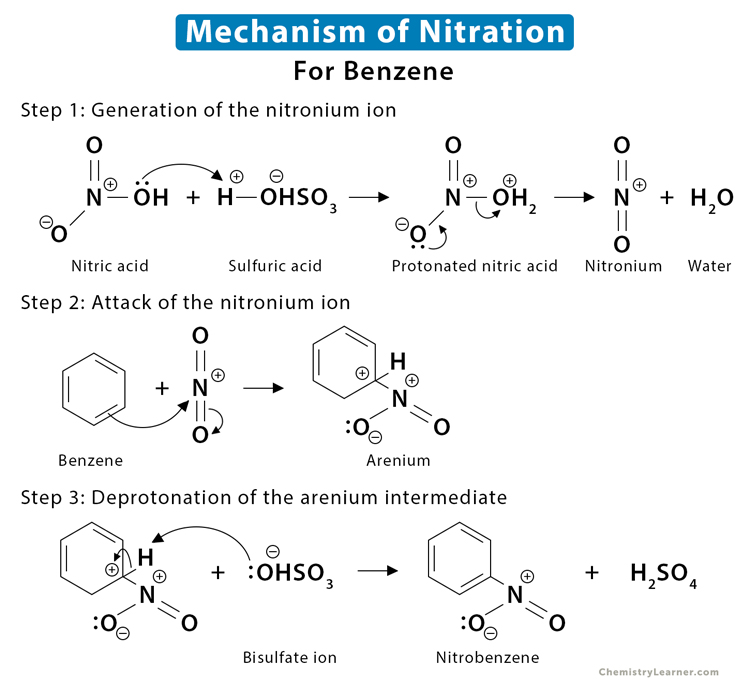
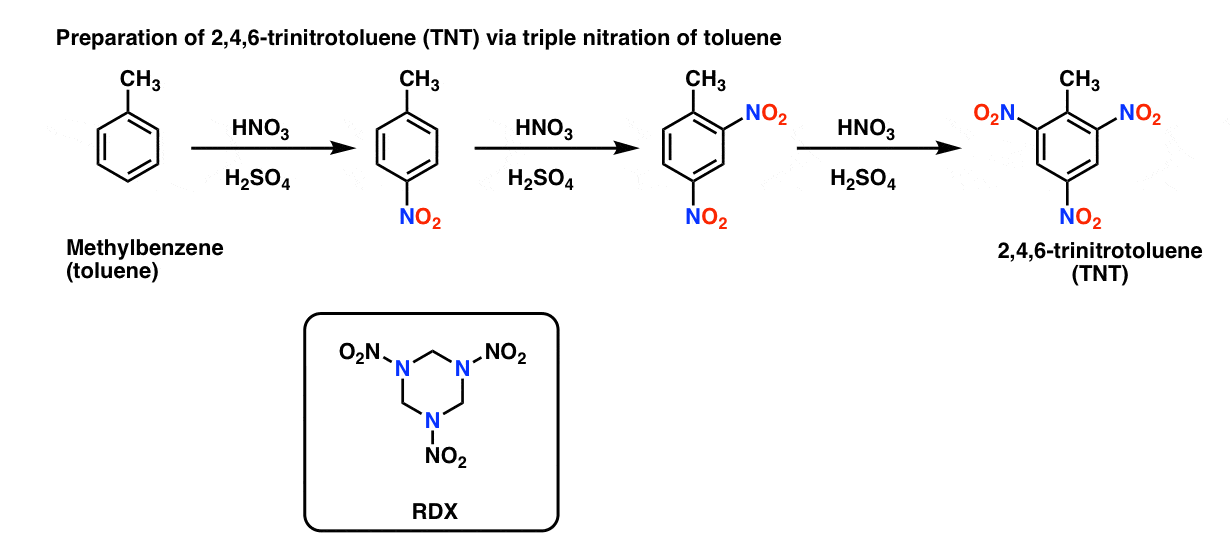
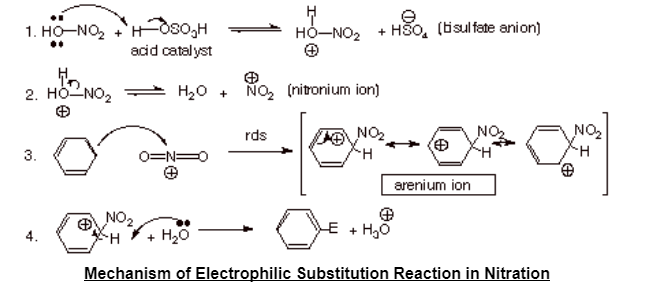
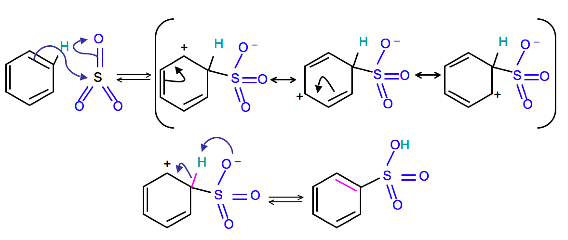
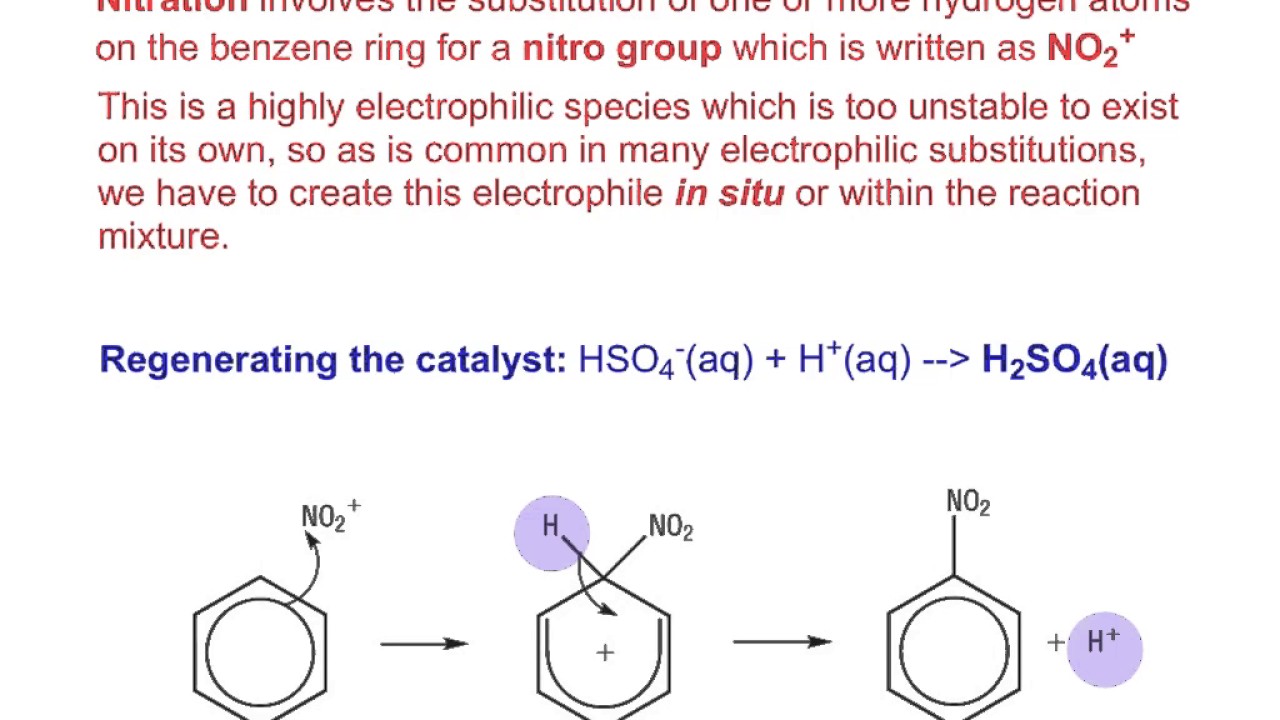
Chemistry lovers - Nitration: A nitro group can be introduced into benzene by using a nitrating mixture to form nitro benzene. The nitrating mixture is a mixture of concentrated nitric acid and
In the nitrating mixture used in the preparation of nitrobenzene from benzene, what does HNO3 act as? - Quora

reaction mechanism - Nitration of tri-substituted benzene with Acetic acid and HNO3 - Chemistry Stack Exchange
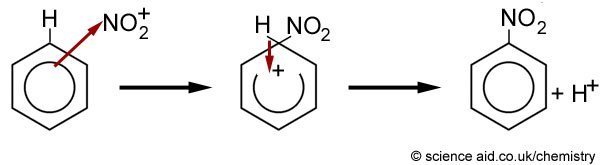
Aromatic Chemistry: Structure of Benzene Ring, Nitration of Benzene, Friedel-Crafts Reactions - ScienceAid