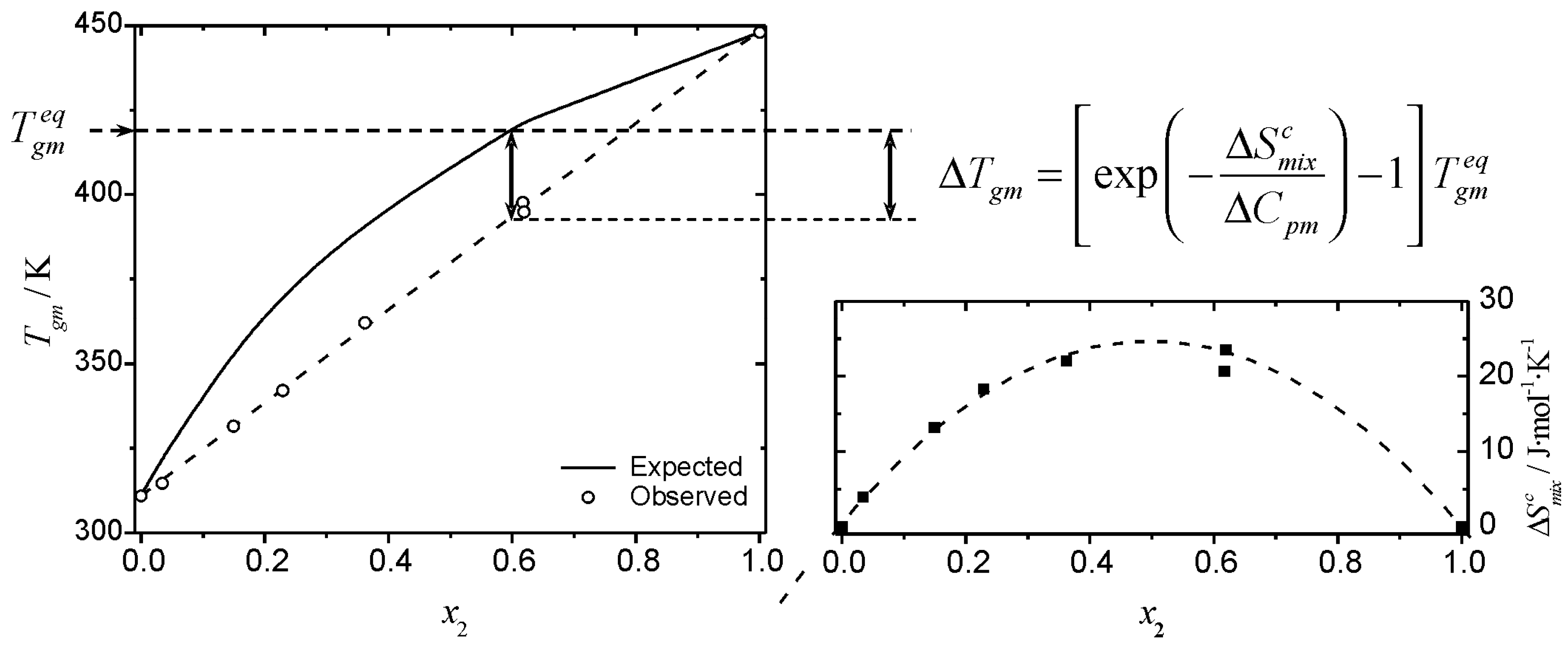
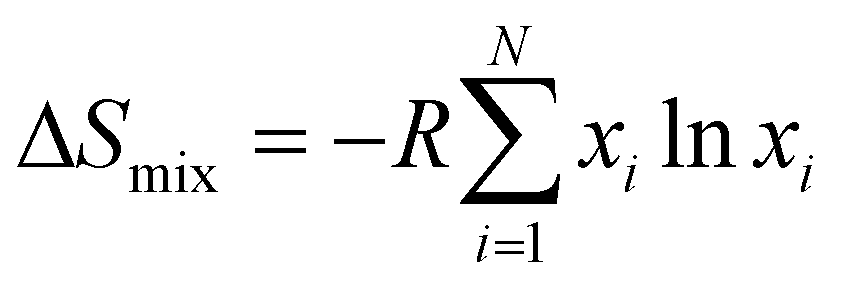The entropy of mixing of the equi-atomic ratio alloys as a function of... | Download Scientific Diagram

Free energy of mixing (a), enthalpy of mixing (b), entropy of mixing... | Download Scientific Diagram

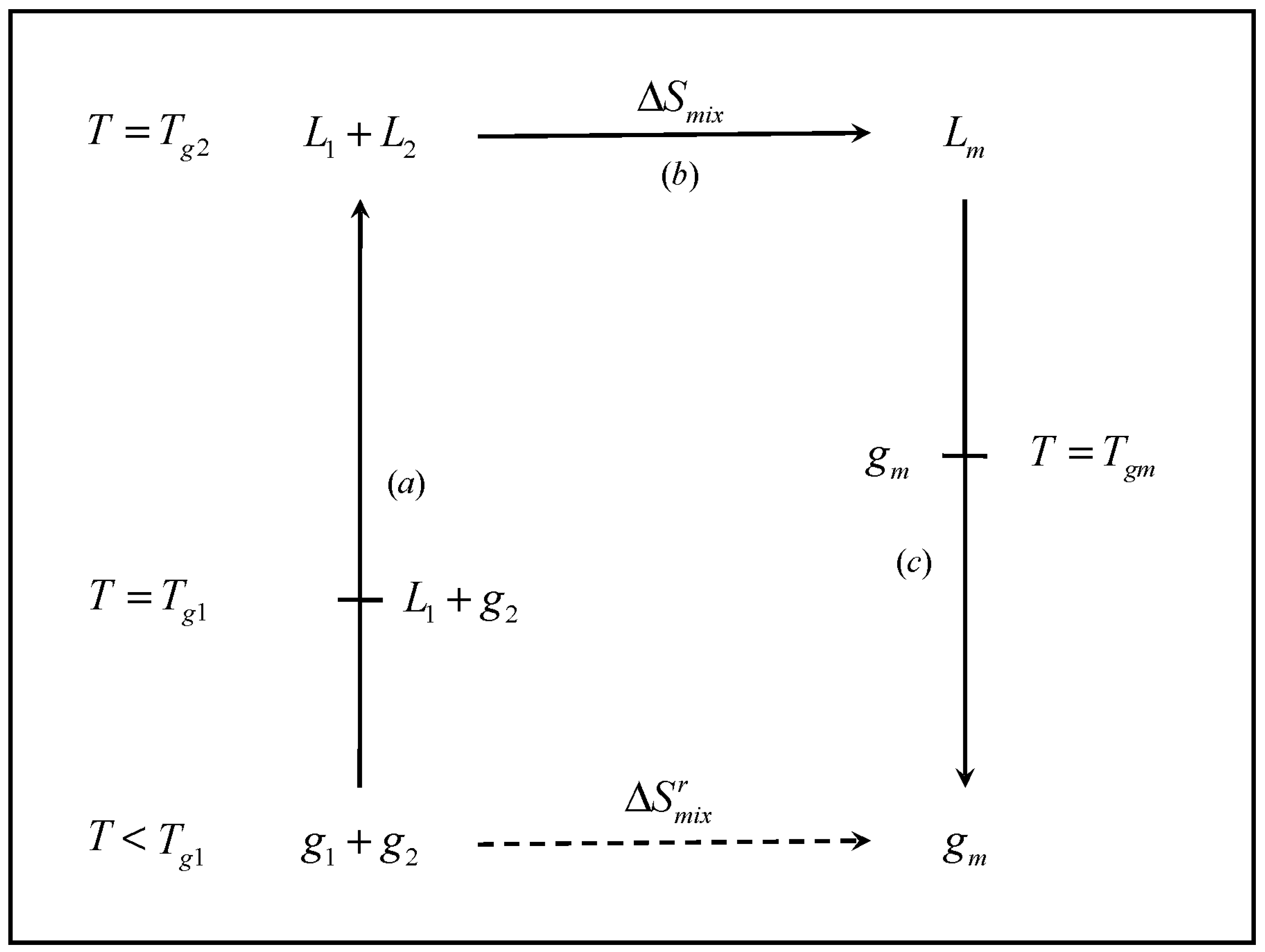
The mixing of entropy, calorimetric, and dynamical behaviors. a ΔH mix... | Download Scientific Diagram

thermodynamics - Determining change in entropy and enthalpy during mixing of gases - Chemistry Stack Exchange
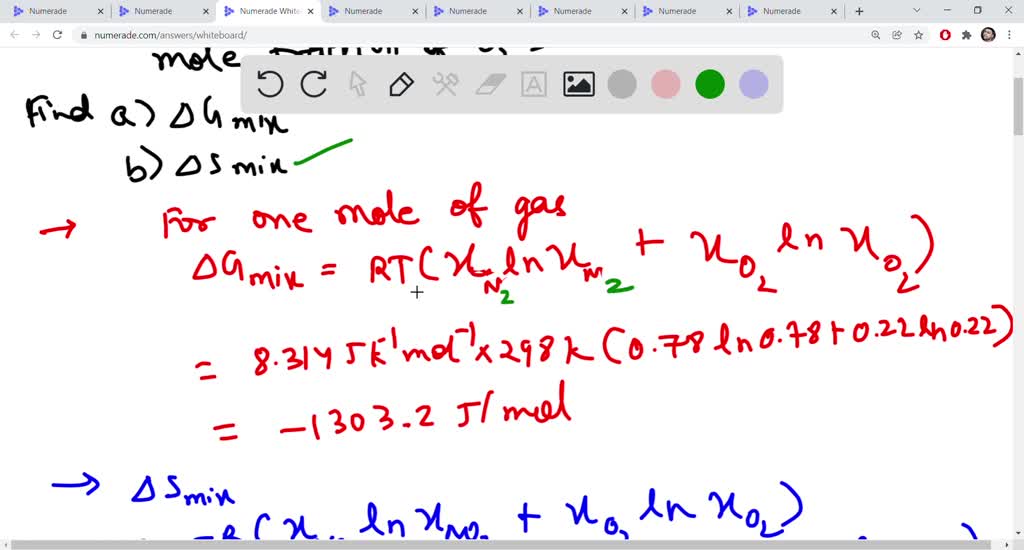
SOLVED: Calculate the (a) molar Gibbs energy of mixing, (b) the molar entropy of mixing when the two major components of air (Nz and 02) are mixed at 298 K The mole
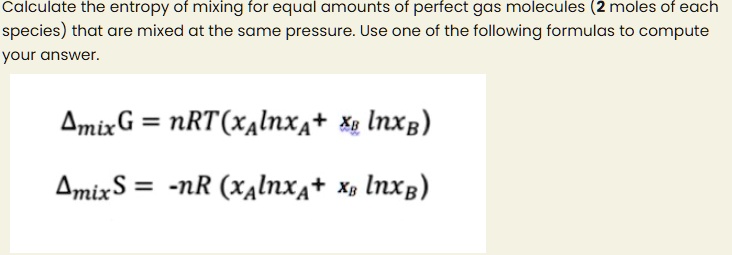
SOLVED: Calculate the entropy of mixing for equal amounts of perfect gas molecules 2 moles of each species) that are mixed at the same pressure: Use one of the following formulas to

Assuming ideal behavior, what is the entropy of mixing (Δ Smix) one mole of N2(g) with one mole of O2(g) at the same temperature and pressure
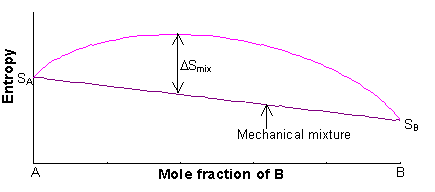
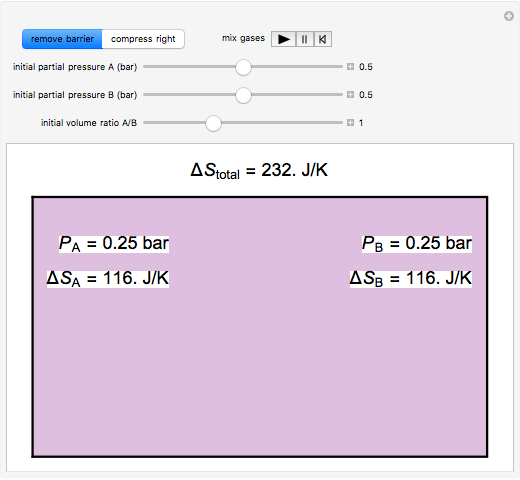
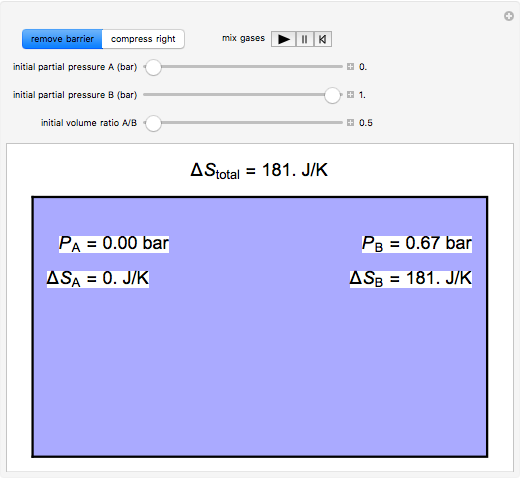
physical chemistry - What is the entropy of mixing of two ideal gases starting out with different pressures? - Chemistry Stack Exchange

The entropy of mixing of 100 g N2O at 120^o C and 1.5 bar with 100 g of CO2 at 120^o C and 1.5 bar is: (log 2 = 0.300)