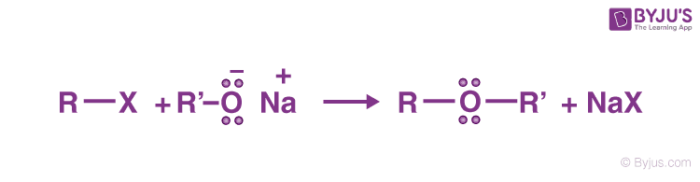Continuous dehydration of ethanol to diethyl ether over aluminum phosphate–hydroxyapatite catalyst under sub and supercritical condition - ScienceDirect
 \\[{\\rm{Al}}{{\\rm{O}}_{\\rm{3}}}\\](C) \\[{\\rm{Cu}}\\](D) \\[{\\rm{Ni}}\\] The formation of diethyl ether from ethanol is catalysed by:(A) \\[{{\\rm{H}}_{\\rm{2}}}{\\rm{S}}{{\\rm{O}}_{\\rm{4}}}\\](B) \\[{\\rm{Al}}{{\\rm{O}}_{\\rm{3}}}\\](C) \\[{\\rm{Cu}}\\](D) \\[{\\rm{Ni}}\\]](https://www.vedantu.com/question-sets/2cc81999-13a6-40db-8255-95464d6521931613393603635429616.png)
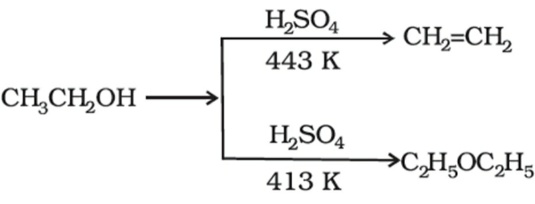
The formation of diethyl ether from ethanol is catalysed by:(A) \\[{{\\rm{H}}_{\\rm{2}}}{\\rm{S}}{{\\rm{O}}_{\\rm{4}}}\\](B) \\[{\\rm{Al}}{{\\rm{O}}_{\\rm{3}}}\\](C) \\[{\\rm{Cu}}\\](D) \\[{\\rm{Ni}}\\]

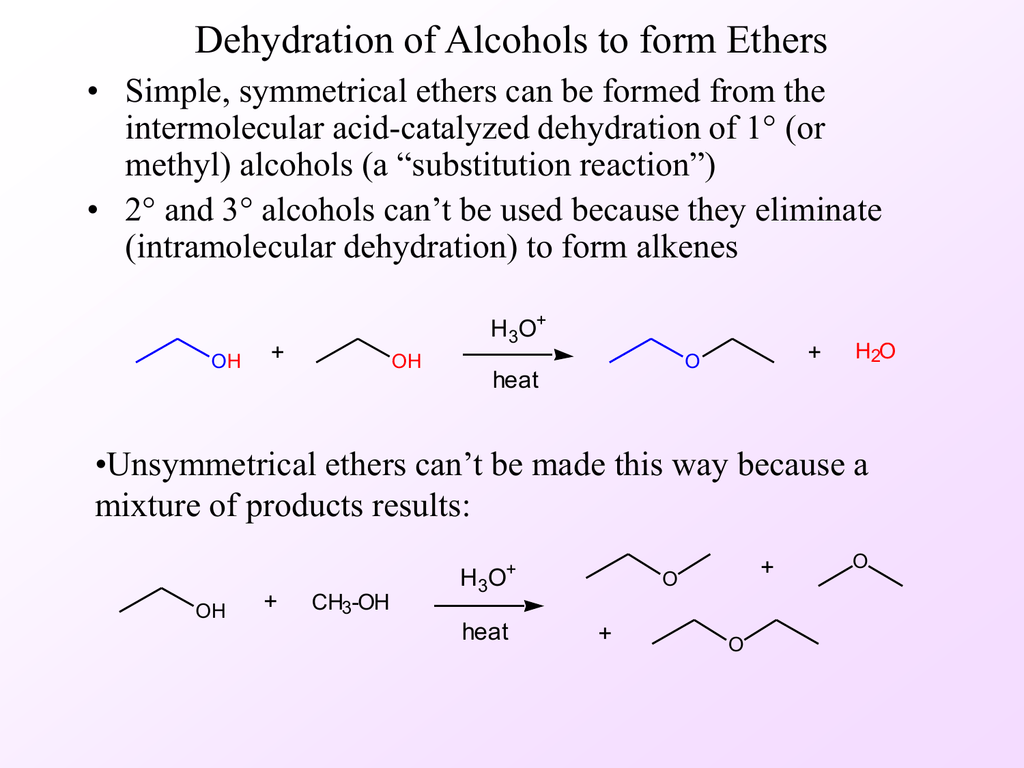
Preparation of ethers by acid-catalysed dehydration of secondary and tertiary alcohols is not a suitable method. Give reason - CBSE Class 12 Chemistry - Learn CBSE Forum

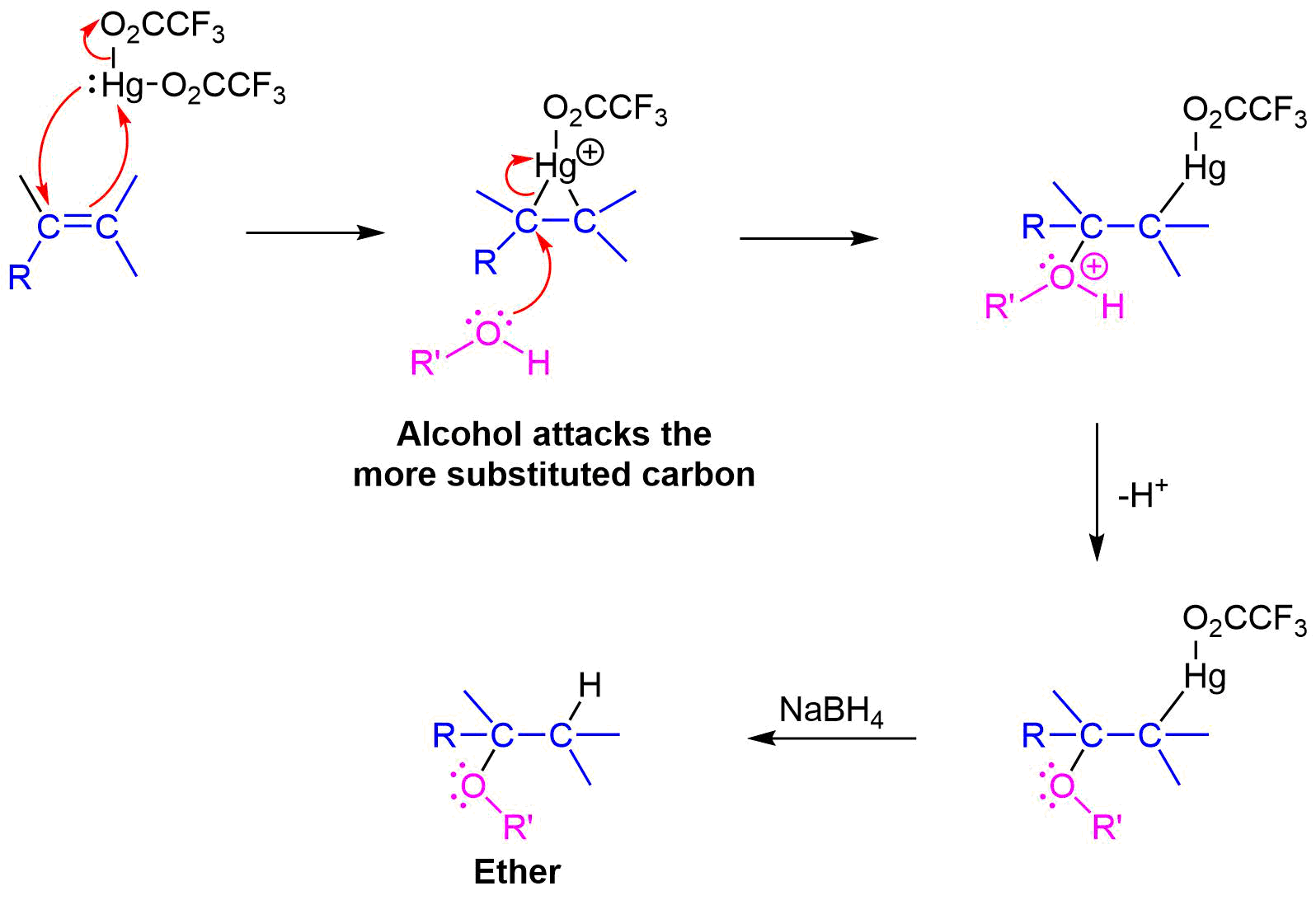
organic chemistry - Why can unsymmetrical tert-butyl ethers be prepared by dehydration of the constituent alcohols? - Chemistry Stack Exchange

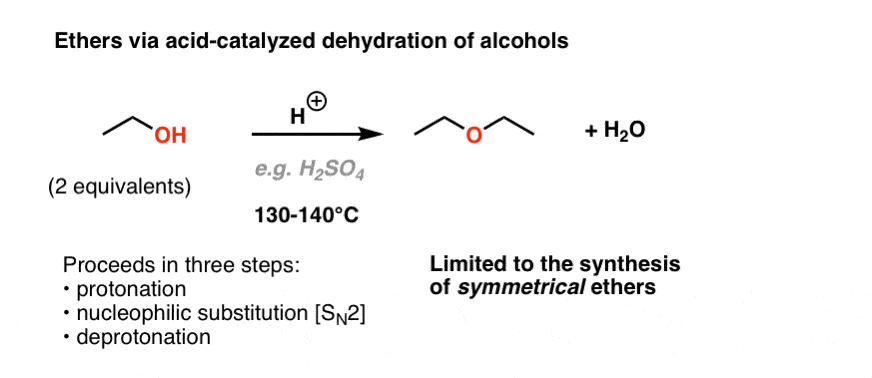
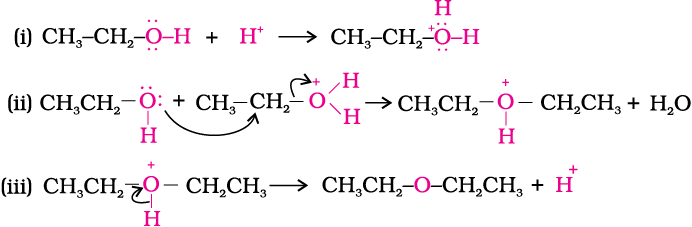
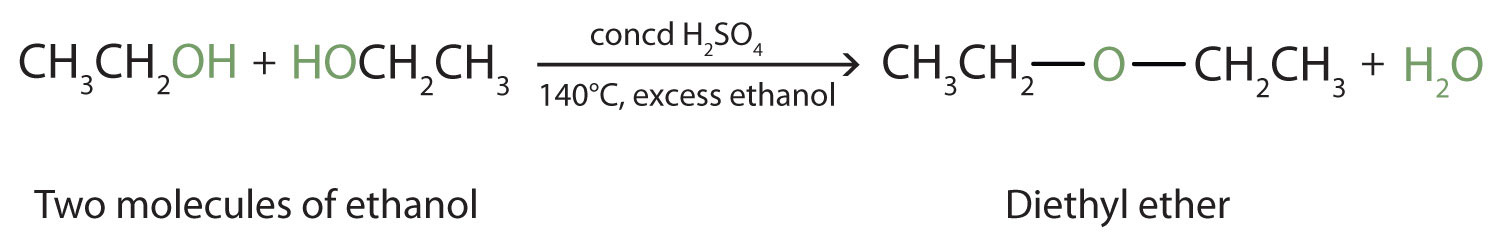
Preparation of ethers by acid dehydration of secondary or tertiary alcohols is not a suitable method

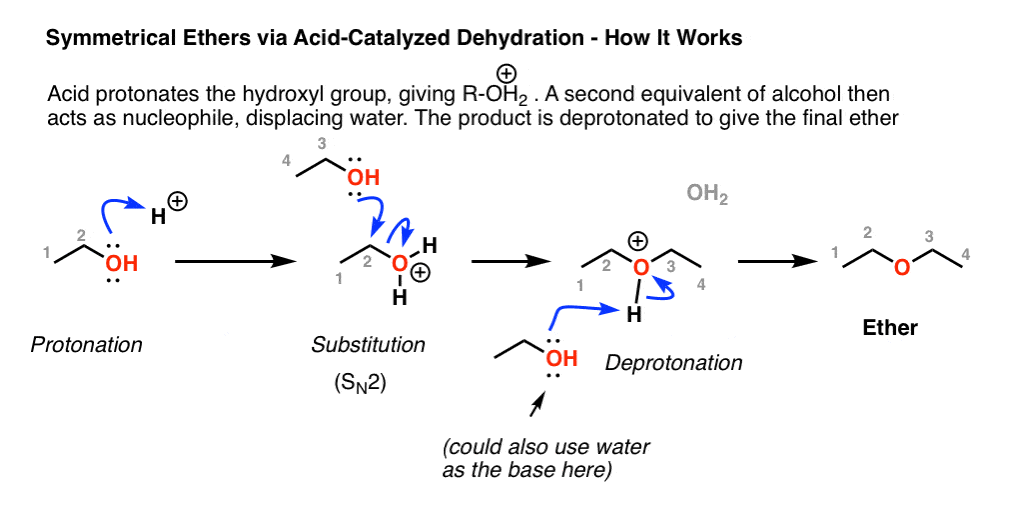
Synthesizing ethers via acid-catalyzed condensation reactions has limited usefulness: the conditions (sulfuric acid and heat) are conducive to elimination products. i. Primary alcohols will undergo d | Homework.Study.com